by Imani Madison
Mitochondria are the powerhouse of the cell, but what happens to cells when they run out of energy? To function, cells need a substantial energy supply in the form of ATP, produced by mitochondria. In addition to ATP, cells also require nutrients and sugars, like glucose, to synthesize macromolecules like proteins, lipids, nucleic acids (e.g., DNA and RNA), and other molecules to support growth, cell division, and overall organism health. To balance energy use with available nutritional and energy resources, cells maintain metabolic homeostasis by switching macromolecular synthesis on and off depending on whether sugars, nutrients, and ATP are plentiful. In humans, when cells can’t properly regulate metabolism, diseases such as diabetes and cancer can develop.
In eukaryotic cells (such as in humans, plants, and animals), protein kinase sensors activate or inhibit metabolism in response to changes in cellular energy, sugar, or nutritional levels. These protein kinases are TORC1 (Target of Rapamycin Complex 1) and AMPK (AMP-activated protein kinase) that cleave a phosphate group from ATP and transfer it to proteins, either activating or inhibiting the now phosphorylated proteins from functioning. When nutrients and sugars are plentiful, TORC1 activates metabolism by phosphorylating proteins and enzymes involved in synthesizing macromolecules through anabolic processes (Figure 1). When nutrients and sugars are scarce, AMPK deactivates metabolism by phosphorylating enzymes and proteins that inhibit TORC1 and promote catabolic processes, which break down molecules to produce energy (Figure 1). This way, cells maintain homeostasis between energy consumption and energy generation.
The TORC1 protein complex contains several proteins that differ between organisms. Across species, the conserved core proteins are: TOR (target of rapamycin), LST8 (lethal with Sec13 protein 8), and RAPTOR (regulatory-associated of TOR). Each protein in the complex has a different function that contributes to the proper functioning of TORC1. TOR is the kinase that performs phosphorylation. LST8 helps TOR phosphorylate proteins effectively. RAPTOR is required for AMPK-mediated TORC1 deactivation. When AMPK is active, it phosphorylates RAPTOR, thereby deactivating TORC1 and preventing it from activating anabolic processes.
TORC1 is especially responsive to changes in sugar levels, especially to glucose. For example, in humans, insulin contributes to high TORC1 activity and to inhibited AMPK activity. Overactive TORC1 may even lead to insulin resistance and type 2 diabetes. TORC1 is also responsive to ATP levels produced by glycolysis as sugars are converted into ATP. As mitochondria generate ATP, TORC1 has high activity in activating metabolism. As sugar and ATP levels rise and fall, cells can rapidly adjust whether to consume energy by synthesizing macromolecules or conserve it by pausing synthetic pathways.
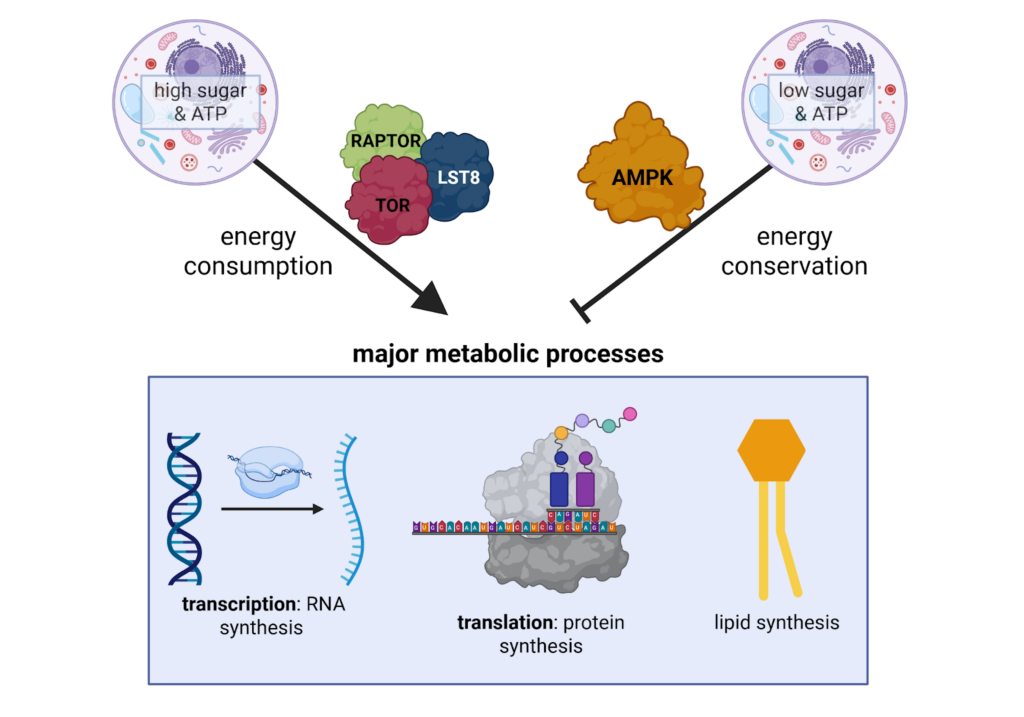
Figure 1 High ATP and glucose (sugar) levels activate TORC1 and promote anabolic processes like protein synthesis, nucleic acid synthesis, and lipid synthesis, while low ATP and glucose levels promote AMPK to inhibit anabolic processes. Figure created in Biorender.com
