by Sarah Angle
Fun Rating: 3/5

Difficulty Rating: 3/5

What is the general purpose? Acids and bases are strong chemicals that can be dangerous to handle and harmful to the environment if not treated properly. The purpose of neutralization is to safely adjust the pH of a solution so it is neither too acidic nor too basic.
Why do we use it? We use neutralization to make acidic or basic solutions safer and more stable. In biology, it helps create the right conditions for cells and enzymes to function properly, since extreme acidity and basicity can damage proteins or halt reactions. It’s also used to safely treat chemical waste before disposal, protecting both people and the environment.
How does it work?
The universal measure of acidity or basicity is called pH, which is measured on a scale from 1 to 14. Things like lemon juice, vinegar, and coffee fall on the acidic side (pH 1–6), while soap, bleach, and milk are more basic (pH 8–14). Right in the middle, at pH 7, is neutral, like pure water.
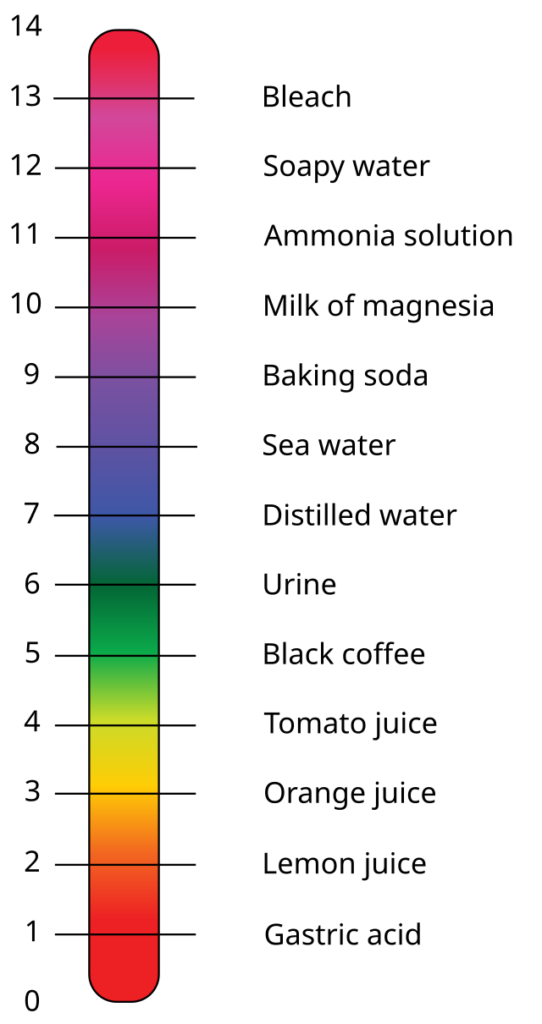
Figure 1. The pH scale shows examples of common acidic, neutral, and basic solutions.
(Stephens, E. 2010. Wikimedia Commons. File:PH Scale.svg|PH_Scale)
In the lab, we aim for that neutral zone because it’s the safest and most natural for living things. To get there, we use a process called neutralization. Acids have extra hydrogen ions (H⁺), and bases have extra hydroxide ions (OH⁻). When they meet, they combine to form water (H₂O), which evens out the pH.
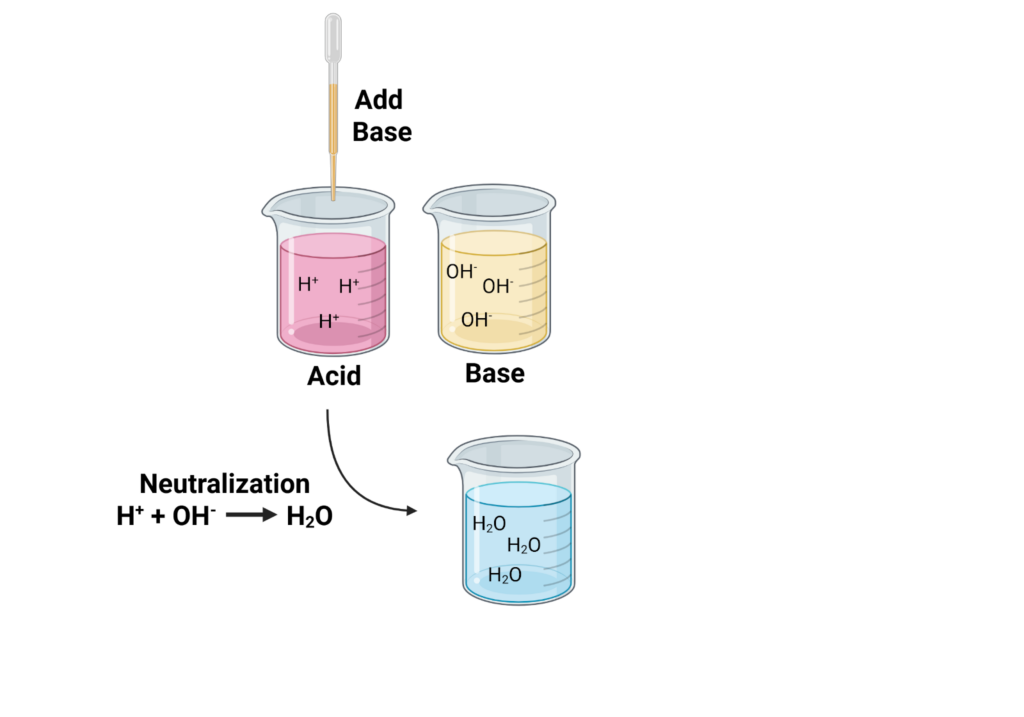
Figure 2. Acids have extra hydrogen ions (H⁺), and bases have extra hydroxide ions (OH⁻). When a base is added to an acid, neutralization occurs, forming water. Image created by author via BioRender.com.
If a solution is too acidic, scientists add a base to raise the pH. If it’s too basic, they add an acid to bring it back down. To keep track, we use colorful pH indicators or digital pH probes to monitor the pH as it moves toward 7.

Figure 3. Antacid tablets react with stomach acid to complete a neutralization reaction.
(Midnightcomm. 2006. Wikimedia Commons. File:Antacid-L478.jpg|Antacid-L478)
Neutralization happens all around us, often without us realizing it. For example, when you have a stomach ache and take an antacid, chemistry is at work! The acid in your stomach reacts with the basic ingredients in the antacid, neutralizing each other. This reaction helps calm the excess acid and makes your stomach feel better.
Check out this video for more on acid and base neutralization!
