by Hazel Milla
You probably know your age based on your birth date. If you’ve read “Epigenetics – More Than What You’re Born With,” you may also know that your DNA can be epigenetically modified so your biology is influenced beyond the genes you inherit. What you may not know, however, is that your age can be predicted based on the epigenetic changes to your genome (aka your epigenome). This is because, as you age, your epigenome ages with you. The age of your epigenome is referred to as your epigenetic age, and how long you have been alive is your chronological age.

Figure 1. A black and white photograph of clocks. Image source.
How is epigenetic age measured?
A common way to measure biological age is with epigenetic clocks. Epigenetic clocks aren’t clocks in the traditional sense. Rather than measuring minutes or hours, they measure how old your epigenome is. They do so using machine learning models, which are algorithms that have been trained to learn patterns of a dataset and use these patterns to predict information about other datasets. Epigenetic aging clocks use DNA methylation microarray data to predict how old a person is based on their epigenome.
How are epigenetic clocks created?
Early biological aging clocks were trained on epigenetic data from people of different ages. They learned DNA methylation levels from portions of the genome relevant to the biology of aging. One commonly used clock, developed by Dr. Steve Horvath, was able to predict people’s ages at 96% accuracy, without ever having seen their data before. Later epigenetic clocks integrated additional data, including protein levels, mortality, and smoking behavior, into their predictions.
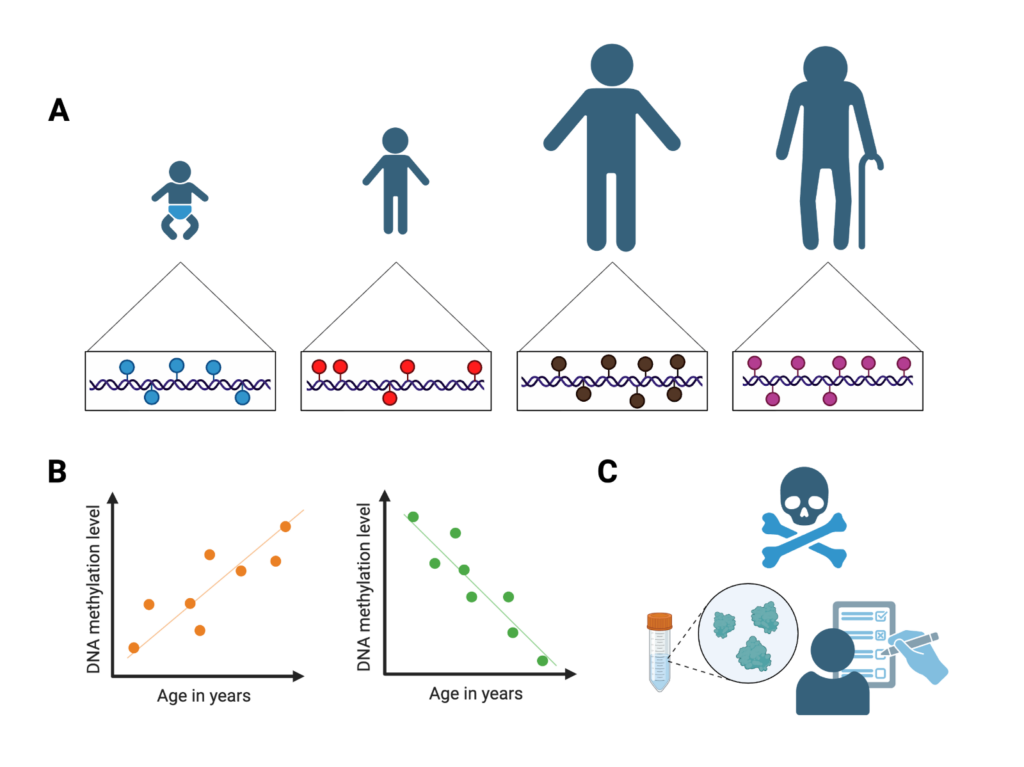
Figure 2. A) Epigenetic clocks are trained using data across different ages to learn age-related patterns of DNA methylation. B) DNA methylation levels correlated with age are used to train epigenetic clocks. C) Later epigenetic clocks also trained their models using additional types of data. Image created by author in Biorender.
How are epigenetic clocks used?
A popular way to use epigenetic clocks is to calculate a person’s epigenetic age and subtract their chronological age from this number. The difference between these two values is referred to as their biological age, a measure that often varies between people. If someone’s epigenetic age is greater than their actual age, we say they have accelerated aging, and if it is less than their actual age, they have decelerated, or slowed-down, aging. We can then run additional analyses to see how differences in biological age predict disease. Accelerated epigenetic age has been associated with cancer, heart disease, and dementia, among other health conditions. Factors that have been linked to accelerated biological aging include childhood stress, lower socioeconomic status, and smoking. Conversely, higher socioeconomic status and healthy habits have been linked to decelerated aging.
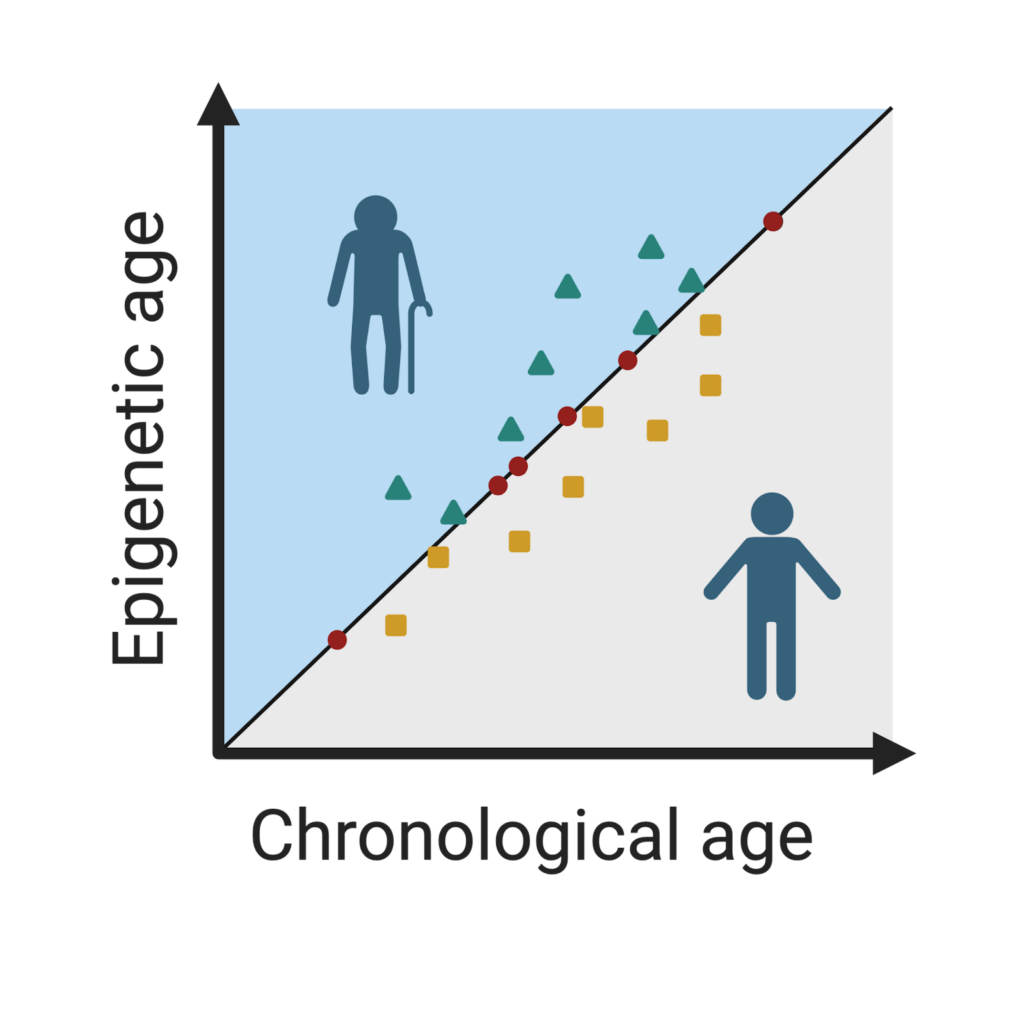
Figure 3. If a person’s epigenetic age is higher than their chronological age (teal triangles), their biological aging is accelerated. If their epigenetic age is less than their chronological age (yellow squares), their biological aging is decelerated. Otherwise, they are aging at an expected pace (red circles). Image created by author in Biorender.
Biological clocks have also been developed using metabolic, transcriptional, and telomeric data, among others. Epigenetic clocks are especially popular given their accuracy and ability to predict disease and mortality for research and clinical purposes. Furthermore, epigenetic age can be modified, meaning that interventions such as a healthy diet, exercise, and meditation potentially slow down biological aging, extending the lifespan.
