by Natalie Thomas
Fun Rating: 4/5

Difficulty Rating: 2/5

What is the general purpose?
Proliferation is a measurement of how rapidly a group of cells divides or replicates, and is an important metric in understanding processes such as growth, development, organ maintenance, and cancer. In a model organism like a mouse, it can be important to identify which cells might be proliferating to better understand the way an organ grows and develops. Suppose we’re interested in learning more about what happens when a specific gene is mutated. In that case, one question we might ask is if that mutation affects proliferation, which can help us better understand its role in diseases like cancer. Ethynyl-deoxyuridine, or EdU, is a tool that allows visualization of proliferation!
Why do we use it?
EdU is a fast, simple, and effective way to identify proliferating cells. In addition to simply identifying proliferating cells, we can use it to determine how fast cells are proliferating, as well as identify senescent cells in culture that are no longer capable of division. EdU is also compatible with immunofluorescent assays, which allow us to detect protein expression of specific genes. This makes EdU a powerful tool and can help us determine which genes might be associated with promoting or blocking proliferation.
How does it work?
Overview
If we look at the full name of EdU, ethynyl-deoxyuridine, it gives us a clue to how it works. The deoxy- in EdU is the same as the “deoxy-” we find in deoxyribonucleic acid, or DNA. Specifically, EdU is a thymidine analog. When a cell proliferates, or divides, it makes a copy of itself. To do this, the cell needs to replicate its DNA through a process called DNA synthesis. When this process occurs, available nucleotides are added to create new DNA. As a thymidine analog, when EdU is present in a cell, it will sometimes be added instead of a normal thymidine nucleotide. We can then utilize a process known as Click-iT chemistry to identify cells in which EdU has been incorporated into the genome. See Figure 1.
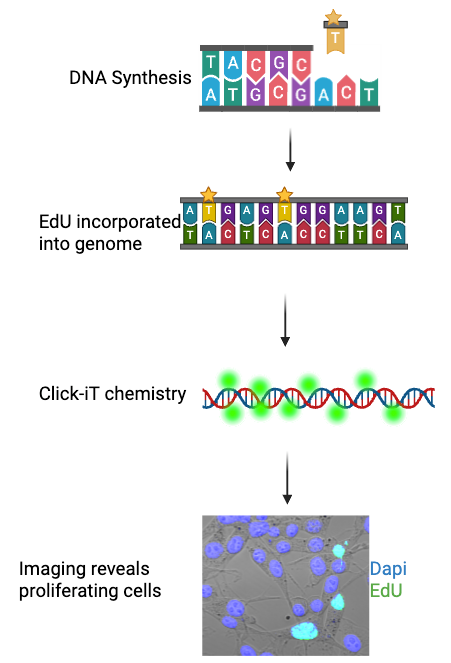
Figure 1: A diagram showing the process of EdU incorporation, Click-iT staining, and imaging showing EdU-positive (green) cells. Dapi, shown here in blue, marks all cell nuclei. Figure generated using BioRender, cell image taken using a Zeiss 880 Confocal Microscope.
Methods of EdU delivery
EdU is a useful tool in both cell culture, where we study the behavior of a specific cell type in a dish, and more complex model systems, such as mice. Introducing EdU to cell culture is extremely easy. By using a 20µM solution of EdU in whatever media you’re culturing your cells in, you can effectively introduce EdU into cells. In small model organisms such as zebrafish or fruit fly larvae, soaking the organism in EdU solution is enough to introduce it into the cells of that organism. For larger animals, such as mice or rats, either an injection or adding EdU to drinking water may be required. To learn more about EdU dosage amounts required for staining, check out this table on ThermoFisher’s website.
Experimental Planning for EdU experiments
There are 3 main types of experiments you might use EdU for. Each is slightly different in the way EdU administration is timed based on the question you might want to answer. These experiments are as follows:
- Instantaneous Labeling: Instantaneous labeling gives information as to what cells are dividing at a specific moment in time. For example, suppose you are interested in learning more about the development of the eye. In that case, you might use instantaneous labeling alongside immunofluorescence of a gene that marks retinal cells to see at what point they are dividing. If you’re studying cancer, it may help you determine which tumors have more proliferating cells than others. In cell culture, it can be used to quantify proliferation in normal cells vs cells where you’ve knocked out a gene. To perform instantaneous labeling, you would expose your cells of interest to EdU for ~2 hours, and then immediately fix or preserve the tissue to prevent further division.
- Cumulative Labeling: Cumulative labeling is useful in situations where your goal is to determine how many cells are capable of proliferating. The differences between cumulative and instantaneous labeling are that in instantaneous labeling, you’ll capture cells actively proliferating, but only the cells that happen to divide in the 2-hour window they’re exposed to EdU. If your cells are only dividing once every 4 hours, this only captures half of the cells capable of division. In cumulative labeling, you expose your cells to EdU for a long period of time, usually several days. After this period, any cells that do not stain positively for EdU are likely cells that are no longer capable of division, or that might be in a quiescent or senescent state.
- Label Retention: A label retention assay is a great tool to determine how quickly cells are dividing and if they are dividing consistently. In this assay, EdU is administered similarly to the instantaneous labeling assay for ~ 2 hours. Instead of immediately fixing the cells, they are then allowed to continue to grow for several days to weeks. As the cells continue to proliferate, EdU becomes more and more dilute, as normal thymidine is still available and added every time the cells divide. This assay might inform us of cells that divide once and then become quiescent, a common feature of stem cell progeny.
