by Claire Greene Whitfield
Fun Rating: 3 / 5
Difficulty Rating: 3 / 5

What is the general purpose?
pH is a chemical property that is key to biological life. It describes the concentration of hydrogen ions [H+] in a substance; the more [H+] ions, the more acidic the solution is. Hydrogen ions are present in everything around us. They enable the chemical reactions necessary for your body to function, including the ones that allow you to read this article right now! Being able to accurately measure pH has enabled scientists to better understand chemistry, biology, and our environment.
Why do we use it?
Maintaining the right pH is essential to health, agriculture, the environment, and modern manufacturing. For example, farmers must maintain a neutral soil pH to grow our food, and we can easily digest that food thanks to the low pH of our stomach acid. The ability to measure the pH of substances, such as blood, water, and soil, allows us to monitor pH levels for changes that could be disruptive or even deadly!
How does it work?
Now that you know how important pH is, how do we measure it? As we discussed, pH is the concentration of hydrogen ions in a substance. To measure pH, we need tools that can detect the number of hydrogen ions present in our sample.
pH is measured on a scale from zero to fourteen, where zero is the most acidic, and fourteen is the most basic (or alkaline). See Figure 1 to find out the pH of some everyday substances.
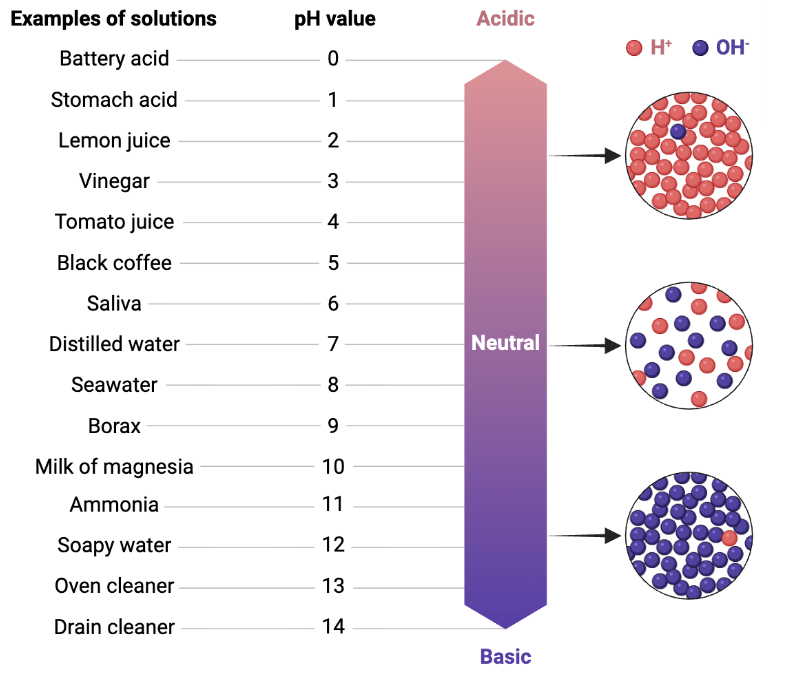
Figure 1. Examples of common solutions on the pH scale. The most acidic solutions have higher concentrations of H+ ions than OH- ions. The opposite is true for the most basic (alkaline) solutions. Notice that water, a neutral solution, has an equal amount of H+ and OH- ions. Figure from Biorender, modified by the author.
How do we measure pH?
There are two main ways to measure pH, depending on how precise you want your measurement to be. You can precisely measure pH (for example, when testing blood pH or making a buffer in a lab) using an electrode. The electrode consists of thin layers of glass and detects the voltage generated by [H+] ions, providing an accurate measure of pH (Figure 2). Another, more old-school method is using litmus paper and a color scale. Just dip the litmus test strip into your solution and use the scale to determine the approximate pH of the sample.
Scientists use these tools to adjust the pH of a substance by neutralizing H+ or OH- ions in a sample. You can read more about how this is done here.
Test your understanding! Observing the right-hand side of Figure 2, what is the approximate pH shown on the litmus paper test strip? (Correct answer at the bottom.)
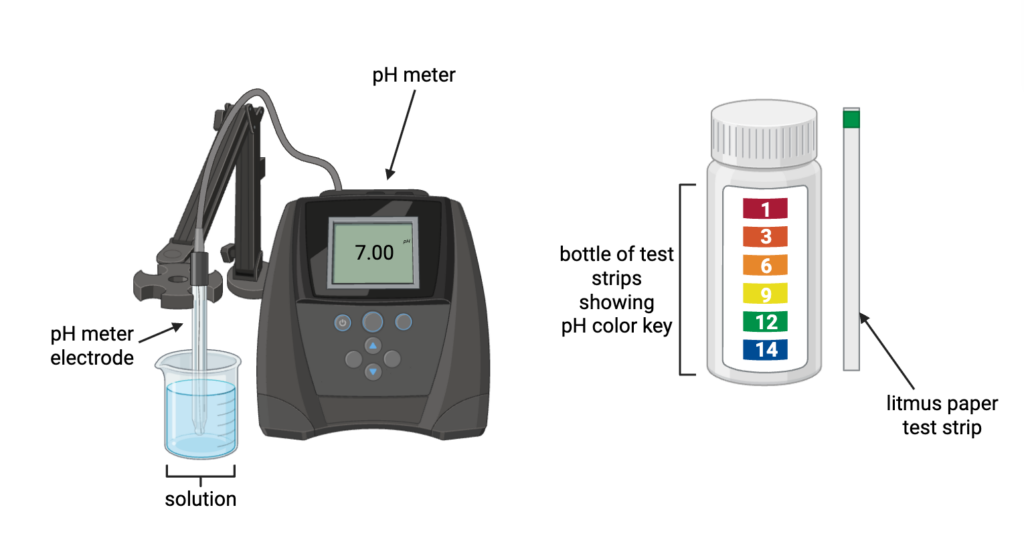
Figure 2. The two main methods of measuring pH are: Using a pH meter provides a precise reading to the decimal point. The litmus paper strategy is quicker but less precise, relying on the user’s best judgment to estimate pH. Figure made by the author in Biorender.
These principles and methods are used daily by scientists across specialties and around the world. Being able to monitor pH helps doctors, farmers, and lab scientists keep our everyday life and health running smoothly.
If you are interested in learning more about how pH impacts health, check out these cool resources:
Correct answer: 12
