by Chloe Gulbronson
Fun Rating: 4/5

Difficulty Rating: 2/5

What is the general purpose?
In Vivo Imaging System (IVIS) Spectrum imaging allows researchers to visualize and track biological processes in living animals in real time. It does so by detecting light signals emitted from cells or molecules of interest.
Why do we use it?
Traditional methods for studying disease progression in animal models require sacrificing groups of mice at multiple time points to see what’s happening inside their bodies. Need to track tumor growth over 8 weeks? That’s potentially dozens of mice. IVIS imaging changes this approach by following the same animals throughout the experiment, allowing biological processes to unfold over time without invasive procedures.
This noninvasive approach is not only more ethical and cost-effective, but it also provides richer data. We can track cancer spread, monitor infections in real-time, evaluate how well a drug reaches its target tissue, and measure treatment responses. This is done while the mouse continues to live a relatively normal lab-mouse life between imaging sessions. Since each animal serves as its own control, this reduces variability and improves the statistical power of the experiments.
How does it work?
Step 1: The Glowing Reporter
The key to IVIS imaging is making our cells of interest visible inside a living animal. We do this by engineering them to produce light, either through bioluminescence or fluorescence.
For bioluminescence, we genetically engineer cells (like tumor cells) to express luciferase, the same enzyme that makes fireflies glow on summer evenings. Before imaging, we inject the mouse with luciferin, the fuel for this reaction. When luciferase encounters luciferin inside the body, a reaction occurs that releases light. The cells will quite literally glow from within!
For fluorescence, we use fluorescent proteins (originally discovered in jellyfish!) that absorb one wavelength (color) of light and emit another wavelength back. The IVIS machine shines the light on the mouse, and the fluorescent proteins will glow in response.
In both approaches, we use an ultra-sensitive camera to track our labeled cells inside a living animal, turning otherwise invisible biological processes into measurable light signals.
Step 2: The Photo Shoot
We use anesthesia to ensure the mouse stays perfectly still during the image capture. Even slight movement can blur the signal, just like in a long-exposure photograph, where any movement ruins the shot.
The anesthetized mouse is placed inside the IVIS imaging chamber, which is essentially a light-tight black box containing an extremely sensitive camera. The chamber blocks out all external light because the camera needs to detect incredibly faint signals coming from deep inside the body, which are scattered and absorbed by layers of tissue, fur, and skin.
The heart of the system is a special detector cooled to -90°C (about -130°F). Why so cold? Cooling reduces electronic noise, allowing the detector to become sensitive enough to detect extremely small amounts of light.
The image acquisition can take anywhere from a few seconds to several minutes, depending on signal strength. Weak signals from small tumors or deep tissues require longer exposure times to collect enough light for a clear image.
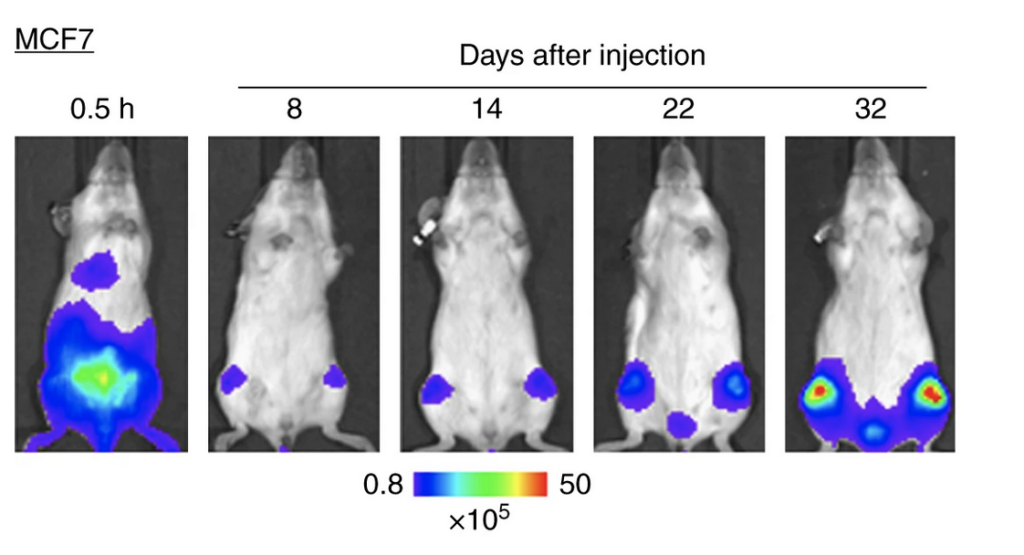
Figure 1: Bioluminescence imaging showing tumor growth over several weeks in the same mouse. Warmer colors (red) indicate higher tumor burden. Adapted from Kuchimaru et al., Nature Communications. 9, 2981(2018) licensed under CC BY 4.0.
Step 3: From Light to Data
After acquisition, the IVIS software processes the raw data into those striking heat-map images you see in Figure 1. The software overlays the light signal onto a standard photograph of the mouse, creating a merged image where warmer colors (red, yellow) indicate stronger signals and cooler colors (blue, purple) indicate weaker signals.
In Figure 1, we can watch breast cancer cells migrate to the bones after injection into mice. Just 30 minutes after injection, we see a green/yellow “hotspot” where the cancer cells are concentrated and initially glowing in the lower abdomen. Over the next few weeks, the signal dims and nearly disappears as the cells travel through the bloodstream and begin settling into the bone. On day 32, the bright red color returns to the mouse’s legs, indicating bone tumors. The cancer cells have spread into the skeleton and multiplied enough to be detected by the imaging system, revealing tumor growth without sacrificing the animal.
But it’s not just about making pretty pictures. The software measures signal intensity and tracks changes over time. This helps us answer questions including: Is the tumor growing? Is the infection spreading? Did the treatment work?
IVIS imaging has transformed how we study disease in living animals by allowing us to observe biological processes unfold in real time rather than piecing together snapshots from different animals. Whether tracking cancer cells as they travel through the bloodstream or watching infections spread through tissue, IVIS imaging brings dynamic biological processes into focus, one glowing particle of light at a time.
